Find Us on the IQVIA COA Marketplace
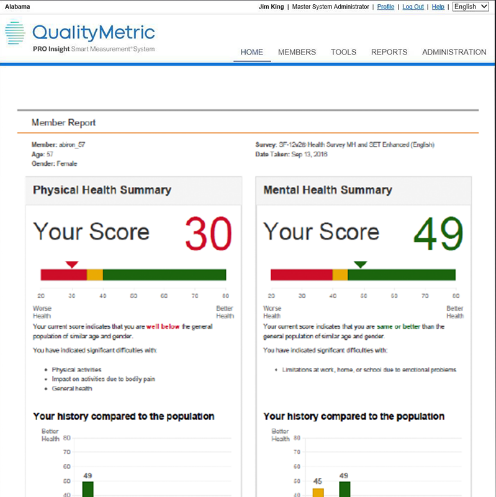
In 2023, QualityMetric (QM) became an IQVIA business. This further enabled us to provide clinical outcome assessment (COA) licensing in addition to leveraging our combined innovative technology solutions and scientific expertise. Since our full integration into IQVIA will soon be complete, it is time to retire the QM brand.
As we remain committed to our same quality licensing and services, we invite you to find the legacy QM COAs you know, in addition to the other 160+ COA offerings on IQVIA's COA Marketplace.